What is Fourier's Law of heat conduction?
q = -kA(dT/dx)
q = kA(dT/dx)
q = kA(T)
q = -k(dT/dx)

Fourier’s Law is a fundamental principle in the physics of heat transfer, stating that the rate of heat flow through a material is proportional to the negative gradient of the temperature and the area through which the heat is flowing. This law is crucial in understanding and predicting how heat moves through different mediums.
Fourier’s Law, a cornerstone in the field of thermodynamics, originates from the work of Jean-Baptiste Joseph Fourier, a French mathematician and physicist. His groundbreaking contributions revolutionized our understanding of heat transfer. Influencing not only physics but also engineering and other sciences.
Fourier introduced his law of heat conduction in the early 19th century, specifically in his seminal work “Théorie Analytique de la Chaleur” (The Analytical Theory of Heat) published in 1822. Through his research, Fourier aimed to describe the flow of heat in solid bodies, developing a theory that heat spreads through solid substrates via a diffusion process.
Fourier’s assertion that the rate of heat transfer through a material. It is proportional to the negative gradient of the temperature field. It was initially met with skepticism. However, his mathematical treatments laid the foundation for what would become the heat equation. Fourier’s methods not only solved practical problems in heat conduction. But also introduced influential mathematical techniques. Such as Fourier series and Fourier transforms. Which are pivotal in various applications across multiple disciplines.
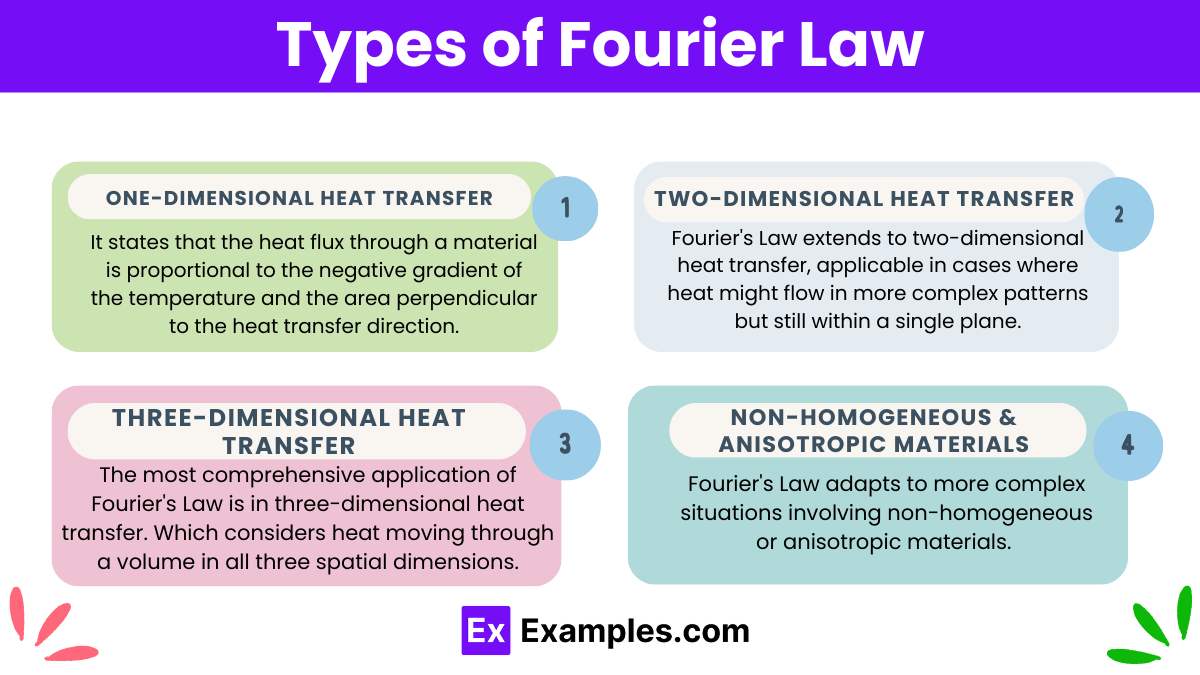
Firstly, we encounter Fourier’s Law in its simplest form in one-dimensional heat transfer scenarios. It states that the heat flux through a material is proportional to the negative gradient of the temperature and the area perpendicular to the heat transfer direction. Typically used in situations involving thin slabs or walls where heat flows in one direction, this formulation is a staple in introductory thermodynamics.
Furthermore, Fourier’s Law extends to two-dimensional heat transfer, applicable in cases where heat might flow in more complex patterns but still within a single plane. Engineers often use this form when analyzing heat transfer across surfaces like building floors or walls, where the heat does not only move straightforwardly but might spread out across the plane of the material.
Moreover, the most comprehensive application of Fourier’s Law is in three-dimensional heat transfer. Which considers heat moving through a volume in all three spatial dimensions. This variant is crucial for designing and analyzing systems where heat transfer is not limited to a single plane. Such as in large machinery, geological formations, or within biological tissues.
Additionally, Fourier’s Law adapts to more complex situations involving non-homogeneous or anisotropic materials. In these cases, the thermal conductivity might vary with direction or position within the material. This adaptation allows for precise predictions and assessments in materials science and engineering, where materials may not uniformly conduct heat due to varying compositions or structured anisotropies.
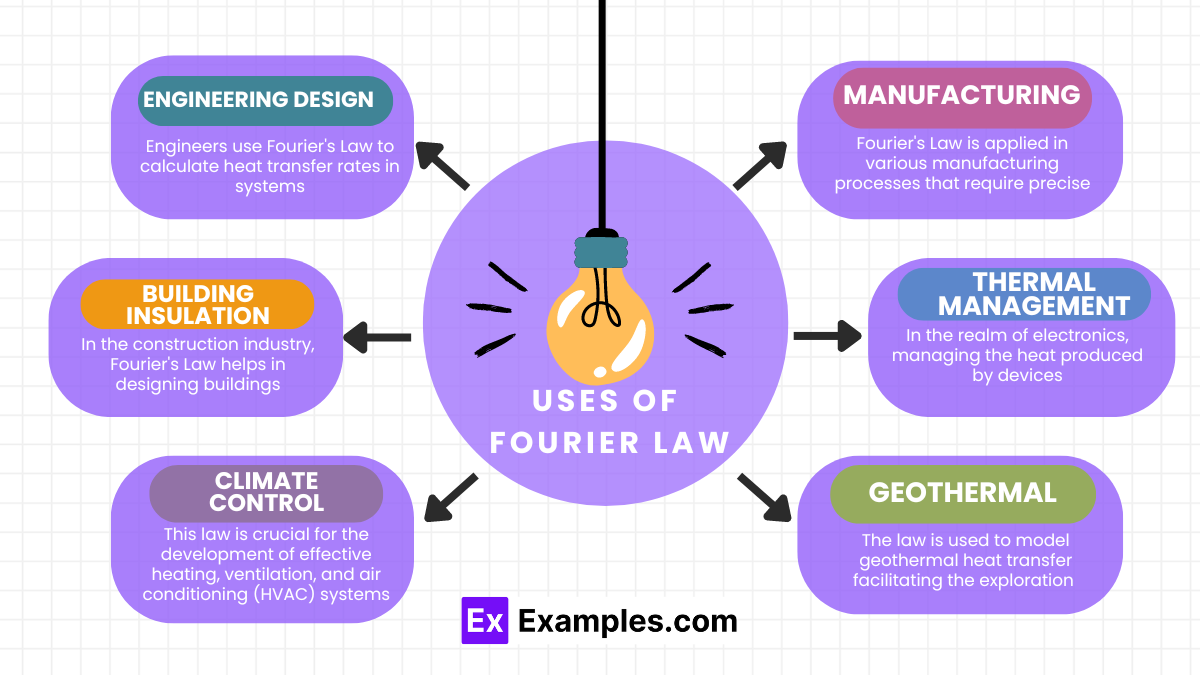
Fourier’s Law is fundamental in the study of heat conduction, providing the basis for calculating how heat moves through materials. This law is particularly crucial for designing efficient thermal systems in engineering and understanding natural heat transfer processes in environmental sciences.
The formula for Fourier’s Law is expressed as:
where:
Fourier’s Law of Heat Conduction is a fundamental principle in thermodynamics that describes how heat transfers through materials. Here’s a step-by-step derivation of this important law:
Before deriving Fourier’s Law, it’s essential to understand some basic concepts in heat transfer:
The derivation starts with the assumption that the heat flux vector (𝑞) is proportional to the local temperature gradient. This assumption is based on observational evidence in steady-state heat transfer (heat transfer at a constant rate over time).
Here, 𝑘 represents the thermal conductivity of the material, a measure of its ability to conduct heat. The negative sign indicates that heat flows from higher to lower temperatures, following the second law of thermodynamics.
Heat Conduction Equation: By considering a differential control volume within the material, you can relate the heat flux to the rate of heat transfer through the material. For a steady-state one-dimensional case in a slab of material with cross-sectional area 𝐴 and thickness 𝑑𝑥, the rate of heat transfer (𝑄˙) can be expressed as:
This equation means the heat flow rate is proportional to the thermal conductivity, the area through which the heat is flowing, and the temperature gradient across the thickness of the slab.
Fourier’s Law can be extended to three dimensions by considering the vector nature of heat flux and temperature gradient:
In this form, 𝑞 and ∇𝑇 are vectors, and 𝑘 may be a scalar or a tensor if the material is anisotropic (i.e., its properties differ in different directions).
To establish a relationship suitable for applying calculus, particularly in varying conditions such as non-uniform materials or temperatures that change over time, we integrate Fourier’s Law with the heat equation. The heat equation in its most general form combines Fourier’s Law with the conservation of energy principle:
Where:
If the thermal conductivity 𝑘 is constant, which is often assumed for isotropic and homogeneous materials, the equation simplifies to:
Here, ∇²𝑇 (or Δ𝑇) is the Laplacian of the temperature. Which gives a measure of the rate at which the temperature field is diverging from any point in space.
Considering the specific heat capacity (𝑐) and density (𝜌) of the material, the heat equation for heat conduction can be written as:
This form relates the change in temperature over time to the spatial distribution of temperature, material properties, and internal heat generation.
To express Fourier’s Law in terms of thermal conductance, consider a simple case of one-dimensional heat flow through a homogeneous material with constant cross-sectional area and thermal conductivity. The formula then simplifies to:
Where:
Steady-state Fourier’s law describes heat conduction when the temperature gradient and heat flow rate remain constant over time, resulting in a stable thermal environment.
Fourier’s law of radiation isn’t a standard term; Fourier’s law specifically describes heat conduction, not radiation. Heat radiation follows Stefan-Boltzmann and Planck’s laws instead.
Fourier’s law is essential for calculating how heat transfers through materials, crucial for designing thermal systems in engineering, building insulation, and various technological applications.
Text prompt
Add Tone
10 Examples of Public speaking
20 Examples of Gas lighting
What is Fourier's Law of heat conduction?
q = -kA(dT/dx)
q = kA(dT/dx)
q = kA(T)
q = -k(dT/dx)
In Fourier's Law, what does the symbol 'k' represent?
Heat flux
Thermal conductivity
Area
Temperature gradient
If the thermal conductivity (k) of a material increases, what happens to the heat flux (q) according to Fourier's Law?
It decreases
It remains the same
It increases
It becomes zero
Which direction does heat flow according to Fourier's Law?
From higher to lower temperature
From lower to higher temperature
It depends on the material
It remains stationary
What does the term (dT/dx) represent in Fourier's Law?
Heat flux
Temperature gradient
Area
Thermal conductivity
If the area (A) through which heat is conducted is doubled, what happens to the heat flux (q) according to Fourier's Law?
It halves
It remains the same
It doubles
It becomes zero
What is the unit of thermal conductivity (k) in the SI system?
W/m²
W/mK
W/K
W/m
In a steady-state heat conduction problem, what can be said about the temperature distribution?
It changes over time
It remains constant over time
It oscillates
It depends on the material
If the temperature difference (dT) across a material is doubled, what happens to the heat flux (q) according to Fourier's Law?
It halves
It remains the same
It doubles
It becomes zero
What role does the negative sign in Fourier's Law (q = -kA(dT/dx)) play?
It indicates the direction of heat flow
It represents heat gain
It signifies thermal resistance
It shows energy production
Before you leave, take our quick quiz to enhance your learning!

